Pure iron, gold and aluminiumĪre too soft for many uses and so are mixed with small amounts of No knowledge of the extraction process of aluminium is required. The properties of copper are limited to its ability to conductĮlectricity easily and the ease with which it can be worked.Īluminium is a useful metal because of its low density and resistance Metals are good conductors of electricity and thermal energy.Ĭopper has properties that make it useful for electrical wiring and 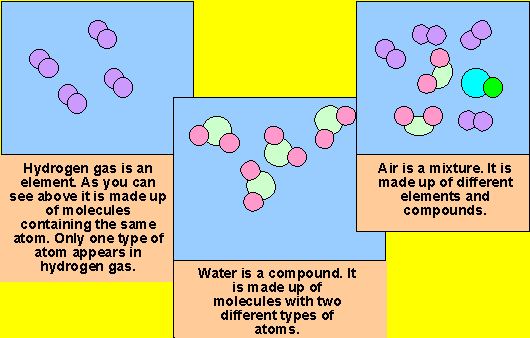
Suggested activity for TDA Compare the properties, such as Metals have giant structures of atoms with strong bonds between theĪtoms and so most metals have high melting points. Students should be able to describe the social, economic andĮnvironmental impacts of mining ores and recycling metals. We should recycle metals to save resources and limit environmental Large amounts of rock need to be quarried or mined to get metal Ores contain enough metal to make it economic to extract the metal. Metals less reactive than carbon can be produced by heating the metal Itself, but most metals are found as compounds that require chemical Unreactive metals, such as gold, are found in the Earth as the metal In inks or food colours using paper chromatography. Suggested activity for TDA Investigate the different colours In paper chromatography a solvent moves through the paper carrying Paper chromatography can be used to separate mixtures and can give Mixtures of water and calcium carbonate that has different particle Suggested activity for TDA Compare the time needed to filter Students should be able to select methods from those given to Mixtures can be separated by processes such as filtration,ĭistillation, crystallisation and chromatography. Students should be able to recognise diamond and graphite fromĪ mixture contains two or more substances not chemically combined Graphite is slippery because the carbon atoms are joined together in Diamond is hardīecause the carbon atoms are joined together in a giant Properties because of their different structures. 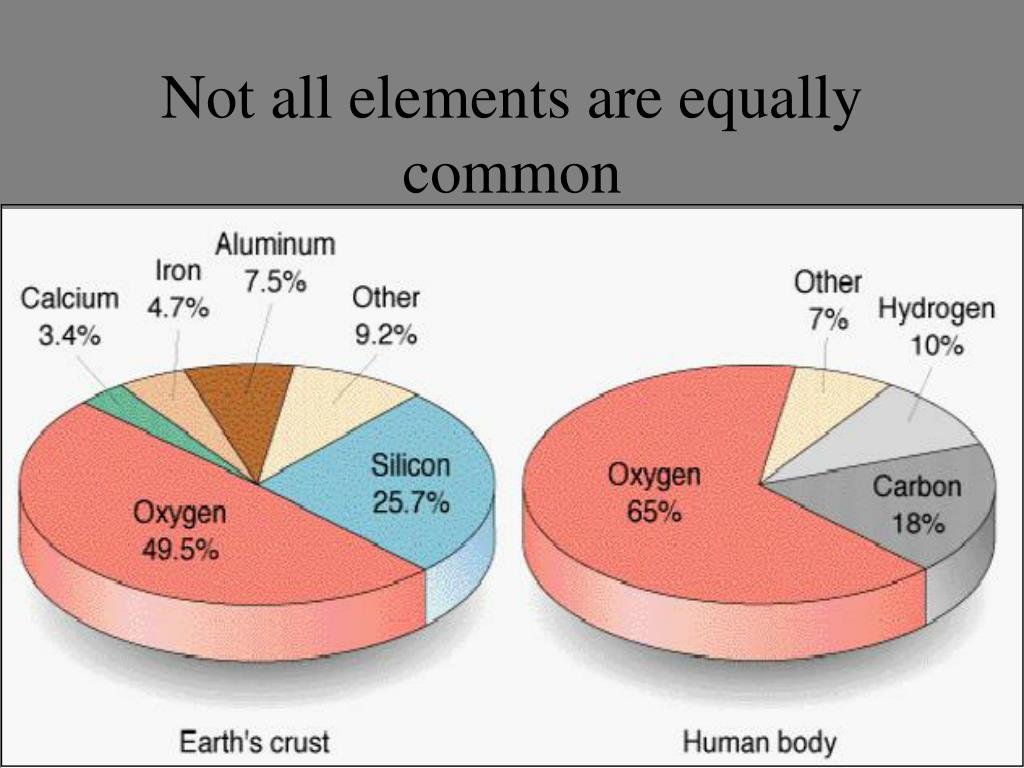
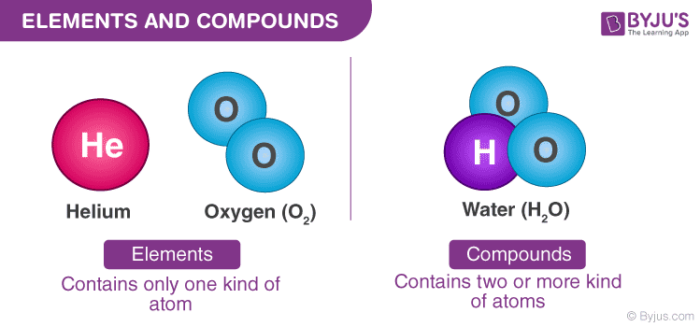
Wax, beeswax polish, butter, margarine, cooking fat.ĭiamond and graphite are forms of the element carbon with different Substances with low boiling points have weak forcesĬompare the melting points of a range different substances, eg candle Melting points have strong forces that hold their particles The particles separate and move about rapidly. When a solid melts to become a liquid the particles are able to moveĪbout but stay close together. Students should be able to use models of particles as small spheres This model, particles are represented by small solid spheres. The three states of matter can be represented by a simple model. Melting andįreezing take place at the melting point, boiling and condensing The three states of matter are solid, liquid and gas. Metals and non-metals, reactions of non-metals to produce oxides,Īnd the other chemical reactions in this specification. Students should be able to write word equations for reactions of Some compounds are made from only non-metals, for example carbonĬhemical reactions can be represented by word equations. Names, eg sodium chloride, magnesium oxide, carbon dioxide. Students should be able to recognise simple compounds from their Some compounds are made from metals combined with non-metals, forĮxample sodium chloride and magnesium oxide. When elements react, their atoms join with other atoms to form Knowledge of groups is limited to Group 1 as reactive metals and Students should know that most of the elements are metals.Įlements in the same group of the periodic table have similar Non-metals towards the right and the top of the periodic table. Metals are towards the left and the bottom of the periodic table and An atom is the smallest part of anĪ substance that is made of only one sort of atom is called anĮlement. Specification reference GCSE Combined Science: SynergyĪll substances are made of atoms. Specification reference GCSE Combined Science: Trilogy Students should have knowledge and understanding of the following content. Mixtures can be separated by processes such as filtration. When two or more elements combine chemicallyĭifferent substances have different combinations of atoms joined together in different ways, which gives them different properties, such as whether they are solid, liquid or gaseous at room temperature. AtomsĪre the building blocks for all substances. Occurring types of atoms called elements.Įlements are shown in the periodic table and are either metals or non-metals. Matter is composed of tiny particles called atoms and there are about 100 naturally Published 15 September 2015 | PDF | 1.1 MB 3.3 Component 3 – Chemistry: Elements, mixtures and compounds
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
